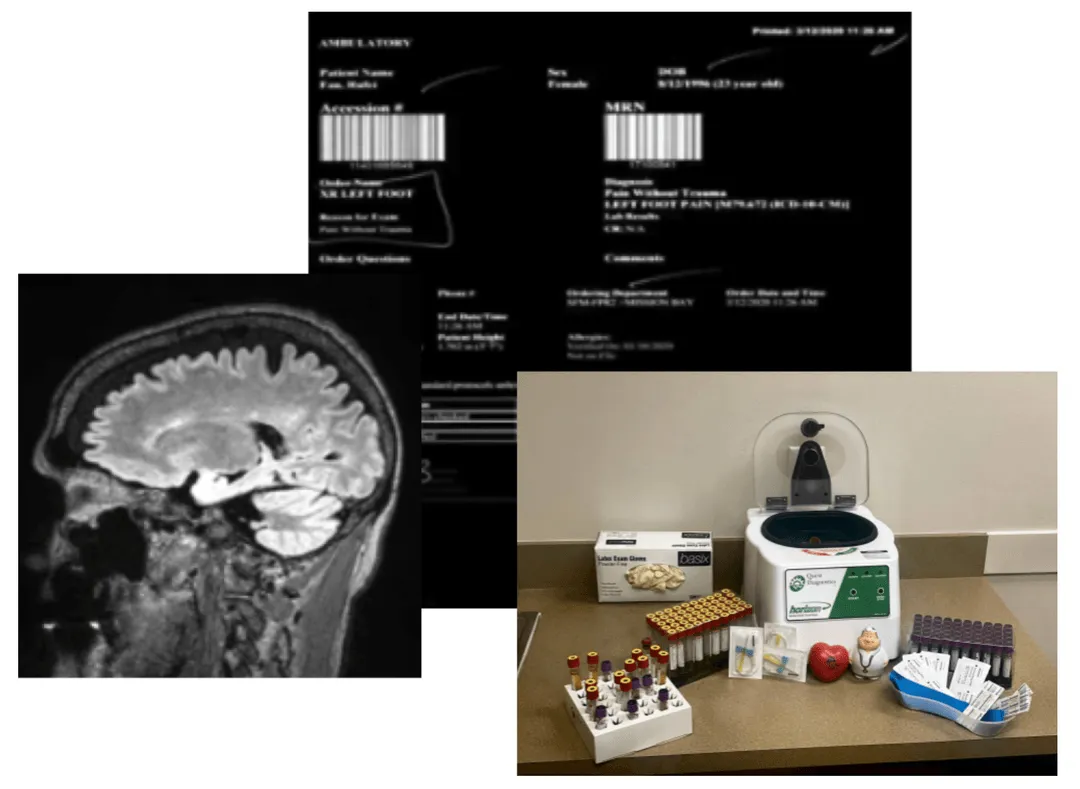
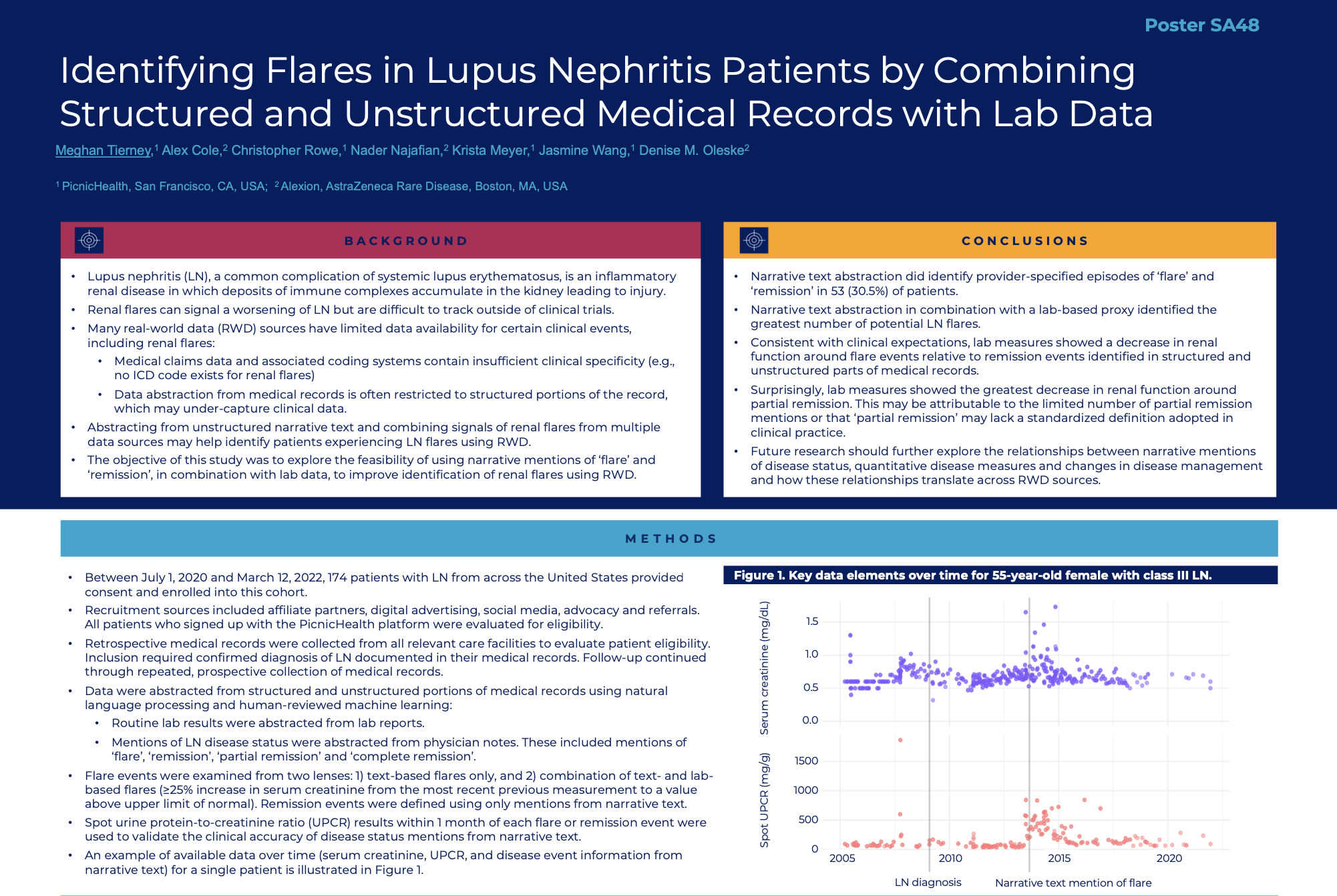
FDA just told industry that it wants to monitor clinical trial data in real time, announcing pilots with AstraZeneca and Amgen.
This is a genuinely exciting development. For decades, critical safety and efficacy signals have moved too slowly from trials to regulators. The gap between phases is not just inefficient. It delays decisions that matter to patients. So the fact that FDA is building infrastructure to close that gap is worth celebrating.
But ensuring real-time access actually gets the right drugs to patients faster means getting a few things right.
The first is independence in who sees what.
Under the current gold standard, accumulating interim data is reviewed by independent Data Safety Monitoring Boards (DSMBs) through monitoring structures that are intentionally separated from sponsors, investigators, and the reviewers who will ultimately assess the submission. That separation is not bureaucratic overhead. It protects judgment from being anchored by incomplete, evolving data.
For real-time regulatory access to preserve that integrity, the system will need a clear trust architecture: who sees what, when, under what charter, and with what separation from the final review team. This may sound like process. It isn't. It is what makes faster decision-making credible.
The second is statistical discipline.
Repeated looks at accumulating data are not neutral. Without pre-specified rules, they can distort inference, particularly when safety, efficacy, or futility signals are being interpreted in flight. DSMBs already operate under analytical constraints: interim analysis plans, alpha-spending rules, and pre-specified stopping boundaries. Real-time regulatory access will need the same rigor applied continuously to ensure decisions are being made with appropriate interpretations of the data.
Underneath both is data provenance.
Real-time access will only be as good as the consent, provenance, structure, and auditability of the underlying data. If the data are fragmented, opaque, or poorly permissioned, "real time" just means faster access to lower-confidence evidence. FDA has consistently emphasized transparency into all procedures, and this practice should continue to extend with flexibility as novel technologies, including AI models, also continue to evolve in real time.
The technology is ready to be used in real time. What actually needs piloting is building an agile trust architecture fast enough to use it wisely and adapt to emerging technologies.
At PicnicResearch, data provenance, auditability, and consent aren't abstract principles for us. They're the foundation of every study we run. As the FDA builds the infrastructure for real-time trial monitoring, we're thinking hard about what that means for the evidence needs of tomorrow. If you are too, let's talk.